|
By installing one of these super-resolution CLSMs at the multi-user Leicester Advanced Imaging Facility, we aim to promote world-class cell biology. These super-resolution CLSMs can come with a detector for improved sensitivity and speed, allowing imaging of live cells where the target molecules may make dynamic movement. CLSMs with super-resolution capability are commercially available. An improved resolution by a factor of 1.7 to 2 (about 100-120 nm) has become the new standard in cell biology. Unsurprisingly, super-resolution microscopy has quickly become very popular in the cell biology research field. Therefore, a question such as whether protein two proteins are co-localising at a particular cellular structure or not, needs to be addressed by super-resolution microscopy. Researchers are aware of this issue and actively working on it but methodology development to precisely "mark" proteins of interest with specialised tags is still ongoing. For example, high resolution imaging by electron microscopy (EM), where resolution can be now down to a few Ånstrongs cannot replace super-resolution microscopy as EM cannot use fluorescent markers and alternative methodologies to localise a protein of interest in relation to the observed structures are limited. The information provided by super-resolution microscopy is unique and cannot be obtained by any other means. 
These microscopes can be called super resolution microscopes (or nanoscopes as they offer nanometre resolution). This major breakthrough in cell biology was awarded the Nobel Prize in Chemistry in 2014 "for the development of super-resolved fluorescence microscopy". In the last two decades, novel methodologies have brought substantial improvements to the resolution, which can become 50-120 nm, dependent on methodologies. Resolution of conventional fluorescence microscopy is limited to about 200 nm, which is termed the "diffraction limit". The quality of images are dependent on their "resolution", which is defined as the minimal distance between two points in the sample that can still be distinguished by the detector (our eyes or a camera) as separate points. This means that both large tissue samples and tiny bacterial samples can be analysed by CLSMs. CLSMs have a flexibility to change the pixel resolution of the image by modulating the pixel setting or zooming into a smaller area of the cell. 
Therefore, we can look inside cells and tissues without physically cutting them into sections and look at specific molecules floating in solution without getting masked by surrounding excess of other molecules. CLSMs allow us to collect signals only from the focal plane, excluding out-of-focus light using a small aperture in front of the detector. These markers are detected by optical microscopes, where a specialised system called Confocal Laser Scanning Microscope (CLSM) has been playing a vital role. They can be used in both fixed samples and in live-cells. 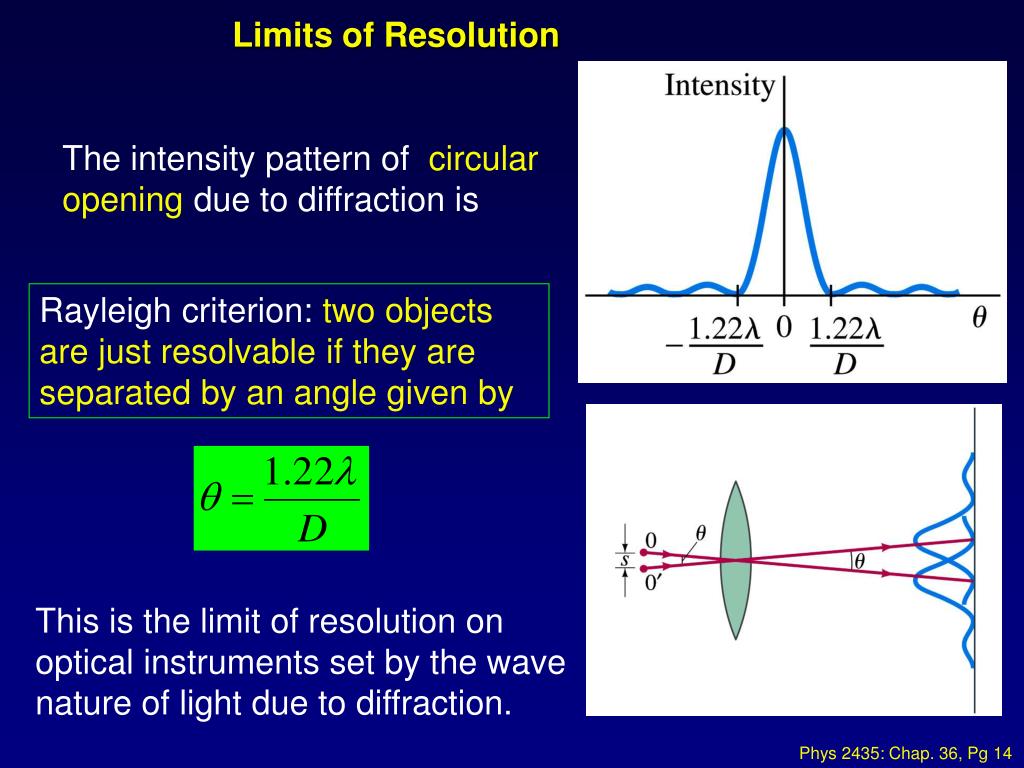
Fluorescent markers that associate with the target molecules have revolutionized our ability to study cellular and tissue developmental and physiological processes. To highlight the locations of the molecules of interest, we need to "mark" the molecules. If these proteins are missing in disease cells/tissues, restoration of these proteins may be an effective therapeutic strategy. If two proteins are found at the same site in a cell, they may work together to deliver a cellular role. For example, if they are found on the cell surface membrane, their role may be relevant to cell membrane function. It is literally "seeing is believing", if we are interested in finding out about microscopic events in a cell/tissue as well as their quantities and dynamic behaviour that will help us to understand their biological roles. Optical imaging of molecules, cells, tissues and whole organisms brings invaluable information when we address biological questions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
